Business model innovation in biotechnology
Dr Swaminathan Subramaniam
Chief Executive - Research Services, Sanmar Speciality Chemicals Limited
 Dr Swaminathan Subramaniam, Swami to his friends, is a man of many parts, someone who combines high academic excellence with unimpeachable professional credentials in the world of business. Graduating from the University of Madras with an MBBS in 1983, he went on to obtain his MD in Clinical Pharmacology from the Postgraduate Institute of Medical Education and Research, Chandigarh. He then pursued doctoral studies in Pharmacology at the University of Pennsylvania, USA, completing his PhD in 1991. The winner of several awards and scholarships, Swami has work experience in both the USA and India in the lifescience/ pharmaceutical sector, starting in laboratory research and moving through a number of business-oriented roles of increasing responsibility, including marketing and business development. His last held position before he joined Sanmar was that of Managing Director and Chief Operating Officer at Aurigene Discovery Technologies Ltd., a Dr Reddy’s company based in Bangalore.
Dr Swaminathan Subramaniam, Swami to his friends, is a man of many parts, someone who combines high academic excellence with unimpeachable professional credentials in the world of business. Graduating from the University of Madras with an MBBS in 1983, he went on to obtain his MD in Clinical Pharmacology from the Postgraduate Institute of Medical Education and Research, Chandigarh. He then pursued doctoral studies in Pharmacology at the University of Pennsylvania, USA, completing his PhD in 1991. The winner of several awards and scholarships, Swami has work experience in both the USA and India in the lifescience/ pharmaceutical sector, starting in laboratory research and moving through a number of business-oriented roles of increasing responsibility, including marketing and business development. His last held position before he joined Sanmar was that of Managing Director and Chief Operating Officer at Aurigene Discovery Technologies Ltd., a Dr Reddy’s company based in Bangalore.

Of the more than 1000 biotech companies floated mostly in the US only a handful are profitable, with the ROI for the rest emphatically in the negative zone. So, why are investors continuing to pour millions of dollars into the biotech sector when experience would indicate that most of them will not see their money back in any form?
What is a biotech company? In India, the term has traditionally been applied to companies that either use microorganisms to produce drugs or apply recombinant DNA technology in the manufacturing process. However, in the US (where most biotech companies are located), the term is applied rather loosely to include any small lifescience company that has an R&D programme with a high-tech biology component.
The origin of the biotech sector boom can be traced back to the 70’s, when (soon after the structure of DNA was determined) the ability to splice together genes from different organisms was demonstrated (the term recombinant refers to this technology). This essentially meant that new organisms with new characteristics could be created in the lab, leading to the fear factor that temporarily put a halt to recombinant DNA experiments. Over time, with better reassessment of the dangers of recombination in the lab, it became possible for
universities and industry to carry out recombination experiments, however under tight supervision.
 A useful byproduct was the opportunity to take a gene, recombine it with a bacterial gene and thereby make the bacterium produce tremendous quantities of the gene product (the protein). Thus protein therapeutics became a reality. This is the technology used by Genentech, Amgen, and homegrown Shantha Biotech, Biocon and Bharat Biotech to produce vaccines and other protein products useful in treating human disease. Proteins are, in some ways, better therapeutic agents than chemicals. Very specific to their targets in the body (a result of their highly complex structure), they usually do not have any unforeseen side-effects, a common problem with chemically based drugs. This also means that the process of getting them to market is simpler, since the regulatory mechanisms are less stringent in terms of the expected toxicity data.
A useful byproduct was the opportunity to take a gene, recombine it with a bacterial gene and thereby make the bacterium produce tremendous quantities of the gene product (the protein). Thus protein therapeutics became a reality. This is the technology used by Genentech, Amgen, and homegrown Shantha Biotech, Biocon and Bharat Biotech to produce vaccines and other protein products useful in treating human disease. Proteins are, in some ways, better therapeutic agents than chemicals. Very specific to their targets in the body (a result of their highly complex structure), they usually do not have any unforeseen side-effects, a common problem with chemically based drugs. This also means that the process of getting them to market is simpler, since the regulatory mechanisms are less stringent in terms of the expected toxicity data.
 So far, so good. But how do you get a protein into the body? Proteins are highly complex and unstable molecules. Acid, heat and the body’s own enzymes (known as proteases) can destabilise them and rapidly render them inactive (albumen in the egg gets denatured when you boil the egg, put lemon juice in milk and it will immediately curdle). Giving proteins orally like a chemical drug, will not work because of the acid and proteases in our gut. So, the only workable alternative is to inject them, a painful prospect in the long-term, as any diabetic on insulin will attest. But scientists do not give up easily and a number of companies have been floated to develop technologies to deliver proteins into the body, long-term and painlessly. Thus, inhaled versions of insulin are just round the corner.
So far, so good. But how do you get a protein into the body? Proteins are highly complex and unstable molecules. Acid, heat and the body’s own enzymes (known as proteases) can destabilise them and rapidly render them inactive (albumen in the egg gets denatured when you boil the egg, put lemon juice in milk and it will immediately curdle). Giving proteins orally like a chemical drug, will not work because of the acid and proteases in our gut. So, the only workable alternative is to inject them, a painful prospect in the long-term, as any diabetic on insulin will attest. But scientists do not give up easily and a number of companies have been floated to develop technologies to deliver proteins into the body, long-term and painlessly. Thus, inhaled versions of insulin are just round the corner.
This limitation also meant that a large bulk of the biotech sector focused on developing traditional, chemical based therapeutics using the tools of biotechnology. Unlike their big pharma counterparts, these mini discovery organisations, were usually biotech start-ups created around ideas that came from university labs. The founders of these ventures—often university professors—would raise venture capital funding from private VCs or the university itself. In the past, academics with ideas for pharmaceutical product development usually went to big pharma with a patent they would license. This was, however not a very good model, as through the 70’s and 80’s, big pharma became enormous bureaucratic organisations lacking the ability to move quickly on radical new ideas.
Entrenched scientists within big pharma also cultivated the Not Invented Here Syndrome. They regarded any ideas from outside as worthless. Academics also were tempted by the opportunity to see their ideas through the initial phases and make some money in the process. Indeed, the success of companies like Genentech and the aura surrounding biotech meant that money was not difficult to come across. VCs would routinely trawl the corridors of academia for bright young academics whose ideas could be funded and then resold for a big profit to a downstream buyer (often big pharma) with biotech stars in his eyes!
 In the US, the biotech sector has grown in the shadows of powerful, dominant pharmaceutical companies such
In the US, the biotech sector has grown in the shadows of powerful, dominant pharmaceutical companies such
as GSK, Pfizer and Merck. Thus many of these companies modelled themselves to produce the discoveries that big pharma was willing to pay big bucks to inlicense. They never saw themselves as marketing companies.
The result was an intense focus on technology and a detachment from the marketplace. And most of these companies were founded by professors who had little idea of (or interest in) how a lab invention got commercialised.
So, the initial wave of companies to attract funding from VCs in perpetual search of the next big thing never became commercially successful. Success in raising funds was useful for some companies who could use their balance sheets to fund subsequent acquisitions or change in focus towards product development. The rest of them, whose timing was not quite right, had to conduct a fire sale of the remaining assets.
Recombinant DNA Technology
Definition: A body of techniques for cutting apart and
splicing together different pieces of DNA. When
segments of foreign DNA are transferred into another
cell or organism, the substance for which they code
may be produced along with substances coded for by
the native genetic material of the cell or organism.
Thus, these cells become “factories” for the
production of the protein coded for by the inserted
DNA.
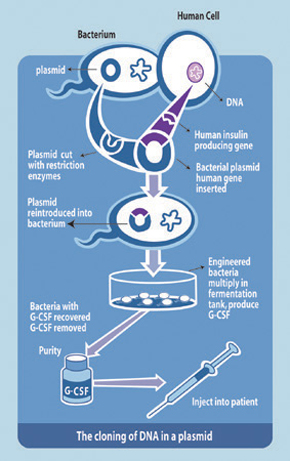
The term “biotechnology” often refers to the techniques of recombinant DNA. This latter term simply refers to the transfer of a gene from one organism into another organism: literally, the recombination of DNA from different sources.
For Amgen’s purposes, this usually involves isolating a human gene with therapeutic potential, then introducing it into bacteria, yeast or an animal cell line. Without recombinant DNA technology, most of these proteins do not exist naturally in sufficient quantities. The recombinant systems, however, can be induced to produce the protein in high quantities under controlled conditions. In the end, we can produce large quantities of a highly purified protein for clinical use.
Source: the website of Amgen, the world’s largest
biotechnology company.